Heat Transfer Miscellaneous
- A long glass cylinder of inner diameter = 0.03 m and outer diameter = 0.05 m carries hot fluid inside. If the thermal conductivity of glass = 1.05 W/mK, the thermal resistance (K/W) per unit length of the cylinder is
-
View Hint View Answer Discuss in Forum
Given data:
d1 = 0.03 m∴ r1 d1 = 0.3 = 0.015 m 2 2
d2 = .05 m
∴ r2 = .025 mK = 1.05 W mK 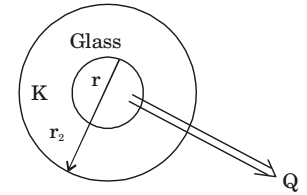
Thermal resistanceRt = 1 loge 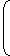
r2 
2πKL r1 = 1 loge 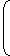
.025 
2 × 3.14 × 1.05 × 1 .015
= 0.077 W/mCorrect Option: B
Given data:
d1 = 0.03 m∴ r1 d1 = 0.3 = 0.015 m 2 2
d2 = .05 m
∴ r2 = .025 mK = 1.05 W mK 
Thermal resistanceRt = 1 loge 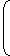
r2 
2πKL r1 = 1 loge 
.025 
2 × 3.14 × 1.05 × 1 .015
= 0.077 W/m
- Consider a long cylindrical tube of inner and outer radii, ri and r0, respectively, length L and thermal conductivity, k. Its inner and outer surfaces are maintained at Ti, and T0, respectively (Ti > T0). Assuming one dimensional steady state heat conduction in the radial direction, the thermal resistance in the wall of the tube is
-
View Hint View Answer Discuss in Forum
Ar = 2πrL
From Fourier ’s Lawqr = kAr dT dr qr = 2π krL dT dr
Boundary conditions:
T = Ti at r = ri
T = T0 at r = r0q = 2πkL(Ti - T0) In(r0/ri) 
Rth = In(r0/ri) 2πkL Correct Option: C
Ar = 2πrL
From Fourier ’s Lawqr = kAr dT dr qr = 2π krL dT dr
Boundary conditions:
T = Ti at r = ri
T = T0 at r = r0q = 2πkL(Ti - T0) In(r0/ri) 
Rth = In(r0/ri) 2πkL
- A hollow cylinder has length L, inner radius r1 outer radius r2, and thermal conductivity k. The thermal resistance of the cylinder for radial conduction is
-
View Hint View Answer Discuss in Forum
In(r2/r1) 2πkL Correct Option: A
In(r2/r1) 2πkL
- For flow of viscous fluid over a flat plate, if the fluid temperature is the same as the plate temperature, the thermal boundary layer is
-
View Hint View Answer Discuss in Forum
NA
Correct Option: D
NA
- An industrial gas (c = 1 kJ/kgK) enters a parallel flow heat exchange at 250°C with a flow rate of 2 kg/s to heat a water stream. The water stream (c = 4 kJ/kgK) enters the heat exchange at 50°C with a flow rate of 1 kg/s. The heat exchange has an effectiveness of 0.75. The gas stream exit temperature will be
-
View Hint View Answer Discuss in Forum

ε = Ch(Th1 - Th2) Cmin(Thi - Tci) 0.75 = 2 ×(250 - Th2) 2 ×(250 - 50)
Th2 = 100° CCorrect Option: B

ε = Ch(Th1 - Th2) Cmin(Thi - Tci) 0.75 = 2 ×(250 - Th2) 2 ×(250 - 50)
Th2 = 100° C

