Heat Transfer Miscellaneous
- In a parallel flow heat exchanger operating under steady state, the heat capacity rates (product of specific heat at constant pressure and mass flow rate) of the hot and cold fluid are equal. The hot fluid, flowing at 1 kg/s with cp = 4 kJ/kgK, enters the heat exchanger at 102°C while the cold fluid has an inlet temperature of 15°C. The overall heat transfer coefficient for the heat exchanger is estimated to be 1 kW/m2K and the corresponding heat transfer surface area is 5 m2. Neglect heat transfer between the heat exchanger and the ambient. The heat exchanger is characterized by the following relation: 2ε = 1 – expt(–2NTU). The exit temperature (in °C) for the cold fluid is
-
View Hint View Answer Discuss in Forum
th1 = 102 °C
tc2 = 15 °CNTU = UA = 1 × 103 × 5 Cmin 1 × 4 × 103 ε = 1 - exp(-2NTU) 2 = 1 - exp(-2.5) = 0.458 2 ∴ 0.458 = tc1 - tc2 = tc1 - 15 th1 - th2 102 - 15
or tc1 = 0.458 × (102 – 15) + 15 = 54.846° C ≈ 55°C
Alternately
C = Mc Cc = MhCh = 1 × 4 kJ/kg K∴ C = Mc Cc = 1 MhCh NTU = U.A. = 1 × 5 = 5 Cmin 4 4
From the relationε = 1 - e-( 2 × 5 / 4 ) = 0.46 2 Now , ε = tc2 - 15 = 0.46 102 - 15
∴ 0.46 = tc2
or tc2 = 54.9Correct Option: B
th1 = 102 °C
tc2 = 15 °CNTU = UA = 1 × 103 × 5 Cmin 1 × 4 × 103 ε = 1 - exp(-2NTU) 2 = 1 - exp(-2.5) = 0.458 2 ∴ 0.458 = tc1 - tc2 = tc1 - 15 th1 - th2 102 - 15
or tc1 = 0.458 × (102 – 15) + 15 = 54.846° C ≈ 55°C
Alternately
C = Mc Cc = MhCh = 1 × 4 kJ/kg K∴ C = Mc Cc = 1 MhCh NTU = U.A. = 1 × 5 = 5 Cmin 4 4
From the relationε = 1 - e-( 2 × 5 / 4 ) = 0.46 2 Now , ε = tc2 - 15 = 0.46 102 - 15
∴ 0.46 = tc2
or tc2 = 54.9
- Saturated vapor is condensed to saturated liquid in a condenser. The heat capacity ratio
is Cr = Cmin . The effectiveness ( ε ) of the condenser is Cmax
-
View Hint View Answer Discuss in Forum
ε of condenser is given by 1-exp(-NTU) Because
Cr = Cmin = 0 ( as Cmax → ∞ ) Cmax Correct Option: D
ε of condenser is given by 1-exp(-NTU) Because
Cr = Cmin = 0 ( as Cmax → ∞ ) Cmax
- One dimensional unsteady state heat transfer equation for a sphere with heat generation at the rate of q can be written
-
View Hint View Answer Discuss in Forum

Correct Option: B

- Two cylindrical shafts A and B at the same initial temperature are simultaneously placed in a furnace. The surfaces of the shafts remain at the furnace gas temperature at all times after they are introduced into the furnace. The temperature variation in the axial direction of the shafts can be assumed to be negligible. The data related to shafts A and B is given in the following Table.
Quantity Shaft A Shaft B Diameter (m) 0.4 0.1 Thermal Conductivity (W/mK) 40 20 Volumetric heat capacity (J/m3K) 2 × 106 2 × 107
The temperature at the centerline of the shaft A reaches 400°C after two hours. The time required (in hours) for the centerline of the shaft B to attain the temperature of 400°C is ____.
-
View Hint View Answer Discuss in Forum
T - T∞ = e(-hAst / ρ V Cp)
(ρ Cp)A = 2 × 106 J/m3K,
(ρ Cp)B = 2 × 107 J/m3K, dA = 0.4 m, dB = 0.1 m
tA = 2hrs = 2 × 3600 = 7200 sec
hA = hB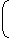
hAs × t 
= 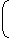
hAs × t 
ρVCp A ρVCp B ⇒ 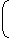
π × 0.4 × L × 7200 
= 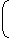
π × 0.1 × L × t × t 
2 × 106 × (π / 4) × (0.4)2 × L A 2 × 107 × (π / 4) × (0.1)2 × L B
⇒ tB = 2.5 hrs
Correct Option: B
T - T∞ = e(-hAst / ρ V Cp)
(ρ Cp)A = 2 × 106 J/m3K,
(ρ Cp)B = 2 × 107 J/m3K, dA = 0.4 m, dB = 0.1 m
tA = 2hrs = 2 × 3600 = 7200 sec
hA = hB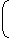
hAs × t 
= 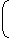
hAs × t 
ρVCp A ρVCp B ⇒ 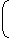
π × 0.4 × L × 7200 
= 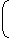
π × 0.1 × L × t × t 
2 × 106 × (π / 4) × (0.4)2 × L A 2 × 107 × (π / 4) × (0.1)2 × L B
⇒ tB = 2.5 hrs
- A cylindrical steel rod, 0.01 m in diameter and 0.2 m in length is first heated to 750°C and then immersed in a water bath at 100°C. The heat transfer coefficient is 250 W/m2K. The density, specific heat and thermal conductivity of steel are ρ = 7801 kg/m3, c = 473 J/kgK, and k = 43 W/mK, respectively. The time required for the rod to reach 300°C is ______ seconds.
-
View Hint View Answer Discuss in Forum
d = 0.01 m
L = 0.2 m, R = 7801 kg/m3
ti = 750 °C, ta = 100 °C
C = 473 J/Kg K, K = 43 W/mK
h = 250 W/m2KLc = V = (π / 4)d2l = d A πdl 4 
T - Ta 
= exp 
-hAτ 
= exp 
-hτ 
Ti - Ta ρVC ρCLc 300 - 100 = exp 
-250 × 4 × τ 
750 - 100 7801 × 473 × 0.01
⇒ τ = 43.49Correct Option: C
d = 0.01 m
L = 0.2 m, R = 7801 kg/m3
ti = 750 °C, ta = 100 °C
C = 473 J/Kg K, K = 43 W/mK
h = 250 W/m2KLc = V = (π / 4)d2l = d A πdl 4 
T - Ta 
= exp 
-hAτ 
= exp 
-hτ 
Ti - Ta ρVC ρCLc 300 - 100 = exp 
-250 × 4 × τ 
750 - 100 7801 × 473 × 0.01
⇒ τ = 43.49

