Refrigeration and Air-conditioning Miscellaneous
- If moist air is cooled by sensible heat removal, which of the following is true?
-
View Hint View Answer Discuss in Forum
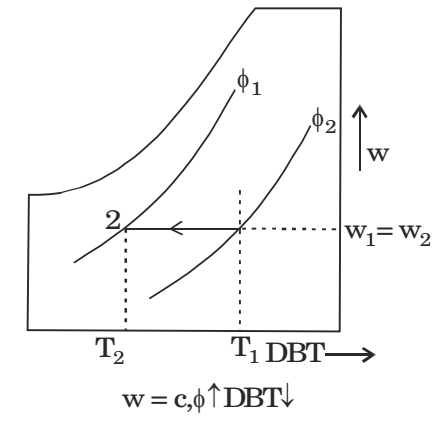
Sensible cooling process
Correct Option: D
Sensible cooling process

- Atmospheric air from 40°C and 60 percent relative humidity can be brought to 20°C and 60 percent relative humidity by
-
View Hint View Answer Discuss in Forum
Cooling and dehumidification process,

Correct Option: A
Cooling and dehumidification process,

- A reversed Carnot cycle refrigerator maintains a temperature of –5°C. The ambient air temperature is 35°C. The heat gained by the refrigerator at a continuous rate is 2.5 kJ/s. The power (in watt) required to pump this heat out continuously is _______
-
View Hint View Answer Discuss in Forum
Q1 = Q2 268 308 Q2 = 2.5 × 308 [Q1 = 2.5] 268
Q2 = 2.873 kW
w = Q2 – Q1 = 0.373 kw = 373.13 Watt.Correct Option: A
Q1 = Q2 268 308 Q2 = 2.5 × 308 [Q1 = 2.5] 268
Q2 = 2.873 kW
w = Q2 – Q1 = 0.373 kw = 373.13 Watt.
- A thin layer of water in a field is formed after a farmer has watered it. The ambient air conditions are: temp. 20°C and relative humidity 5%. An extract of steam tables is given below.
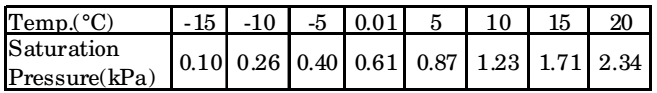
Neglecting the heat transfer between the water and the ground, the water temperature in the field after phase equilibrium is reached equals
-
View Hint View Answer Discuss in Forum
Given data:
Φ = 5% = 0.05
Tdb = 20°C
From table
At 20°C, Ps = 2.34 kPaΦ = Pv Ps 0.05 = Pv 2.34
Pv =.117 k Pa By Interpolation method,Tdb = - 15 + (-10 + 15 × (0.117 - .1) 0.26 - 0.10
= – 15 + 0.531 = – 14.5° CCorrect Option: C
Given data:
Φ = 5% = 0.05
Tdb = 20°C
From table
At 20°C, Ps = 2.34 kPaΦ = Pv Ps 0.05 = Pv 2.34
Pv =.117 k Pa By Interpolation method,Tdb = - 15 + (-10 + 15 × (0.117 - .1) 0.26 - 0.10
= – 15 + 0.531 = – 14.5° C
- A heat engine having an efficiency of 70% is used to drive a refrigerator having a coefficient of performance of 5. The energy absorbed from low temperature reservoir by the refrigerator for each kJ of energy absorbed from high temperature source by the engine is
-
View Hint View Answer Discuss in Forum
Given: ηengine = 0.7, (C.O.P)R = 5
ηengine = W ...(i) Q1 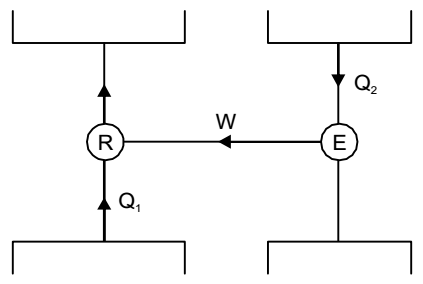
Now,(C.O.P)R = Q2 W ∴ W = Q2 ...(ii) 5
Again,0.7 = Q2 × 1 5 Q1 or Q2 = 3.5 Q1
Energy absorbed from low temperature reservoir by the refrigerator for each kJ of energy absorbed from high temperature source by the engine = 3.5 kJ.Correct Option: C
Given: ηengine = 0.7, (C.O.P)R = 5
ηengine = W ...(i) Q1 
Now,(C.O.P)R = Q2 W ∴ W = Q2 ...(ii) 5
Again,0.7 = Q2 × 1 5 Q1 or Q2 = 3.5 Q1
Energy absorbed from low temperature reservoir by the refrigerator for each kJ of energy absorbed from high temperature source by the engine = 3.5 kJ.

