-
An electron of a stationary hydrogen atom passes from the fifth energy level to the ground level. The velocity that the atom acquired as a result of photon emission will be :
(m is the mass of the electron, R, Rydberg constant and h Planck’s constant)
-
-
24hR 25m -
25hR 24m -
25m 24hR -
24m 25hR
-
Correct Option: A
For emission, the wave number of the radiation is given as
| = RZ2 | 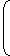 | - |  | |||||
| λ | n21 | n22 |
R = Rydberg constant, Z = atomic number
| = R | 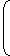 | - |  | 12 | 52 |
| = R | 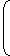 | 1 - |  | 25 |
| ⇒ | = R | |||
| λ | 25 |
linear momentum
| P = | = h × R × | |||
| λ | 25 |
(de-Broglie hypothesis)
| ⇒ mv = | ⇒ V = | |||
| 25 | 25m |

