-
The wavelength of the first line of Lyman series for hydrogen atom is equal to that of the second line of Balmer series for a hydrogen like ion. The atomic number Z of hydrogen like ion is
-
- 3
- 4
- 1
- 2
Correct Option: D
For first line of Lyman series of hydrogen
| = Rhc | 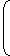 | - |  | |||||
| λ1 | 12 | 22 |
For second line of Balmer series of hydrogen like ion
| = R2hc | 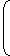 | - |  | |||||
| λ2 | 22 | 42 |
By question, λ1 = λ2
| ⇒ | 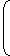 | - |  | 1 | 2 |
| = Z2 | 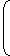 | - |  | or Z = 2 | 4 | 16 |

